Micro Transfer Printing Technology Applied to Silicon Photonics – Why?
Photonics Light the Way
The demand for data is never-ending and instantaneous access is the goal to meet. With the coming of Artificial Intelligence (AI), Machine Learning (ML), Quantum computing, and data centers there is a challenge to meet the connectivity demands of billions of devices. Photons move at the speed of light without the interference that comes from electronic transmission.
With photons, data is transmitted faster and more efficiently, with lower latency and using less power.

Smartphones, tablets, wearables, automotive sensors, IoT devices and computers all rely on photonic chips. Components are combined to expand the functionality of the chip without increasing the size, using minimal packaging and producing less waste.

Heterogeneous Integration & Photonics
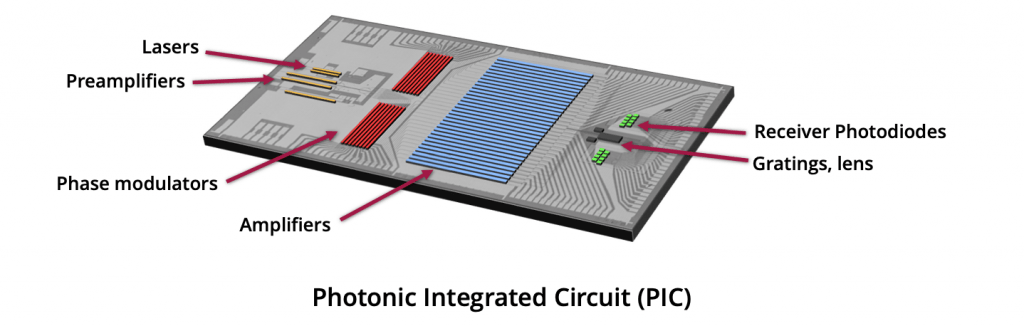
Using heterogeneous integration multiple tiny components – laser diodes, modulators, photo detectors, optical amplifiers, and optics – are precisely aligned on a single substrate, with wave guides on a wafer.
- Efficient assembly of multiple heterogeneous materials
Lasers, Modulators, Detectors, Driver ICs: analog and digital. - Precision alignment at each step
Efficient coupling of devices to waveguide wafers - Mass Transfer
Scales to high device count as performance demands continue to rise - Minimized device sizes
Significant savings of expensive III-V materials
X-Celeprint at Upcoming Conferences

Photonics West 2026

OFC 2026

IEEE CICC 2026
What X-Celeprint Offers
We empower stakeholders to meet the demand for Faster, Smaller, higher Performance, Energy Efficient and Environmentally friendly components through heterogeneous integration.
Based on our unique and extended MTP patent portfolio that is licensed, we support our clients with feasibility, prototyping and industrialization guided by our MTP experts and access to the X-Celeprint eco-system.
It’s our job to provide the expertise and license IP to enable our clients to produce a high-quality Photonics Integrated Circuit (PIC) that delivers for their customers and vendors.
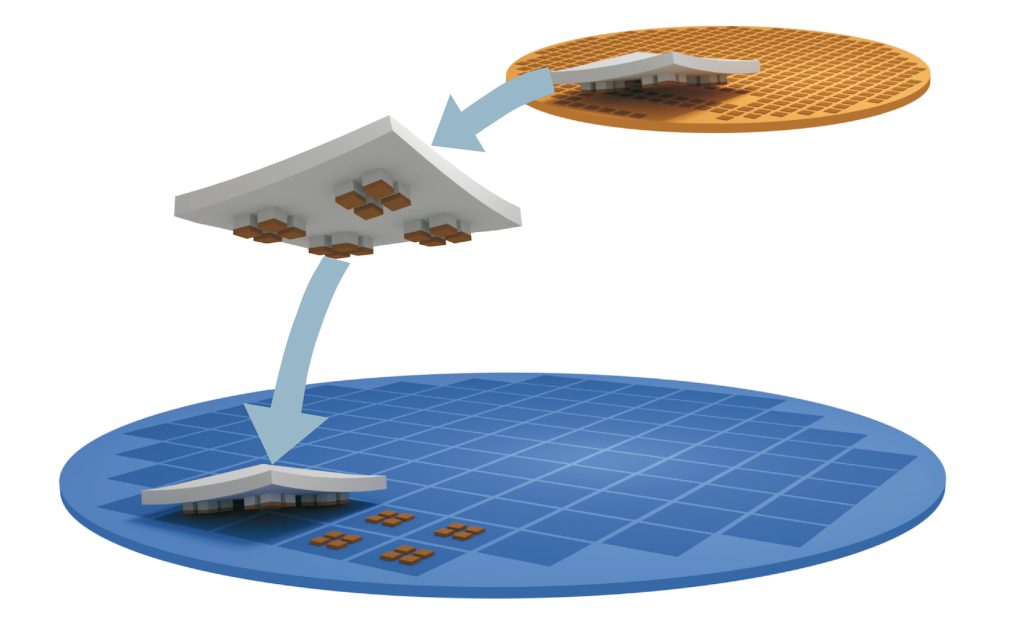

X-Celeprint is an international leader in micro-transfer printing. Our headquarters are at the Tyndall National Institute in Cork, Ireland. We also have a Development Center in Research Triangle Park, NC. We serve customers in North America, Europe, and the Asia Pacific. We provide our clients with design support, prototyping, printing, licensing, and tech transfer for commercialization.